CorLog – Continuous hemodynamic monitoring in the right heart

continuous

30 days

wireless

easy to remove
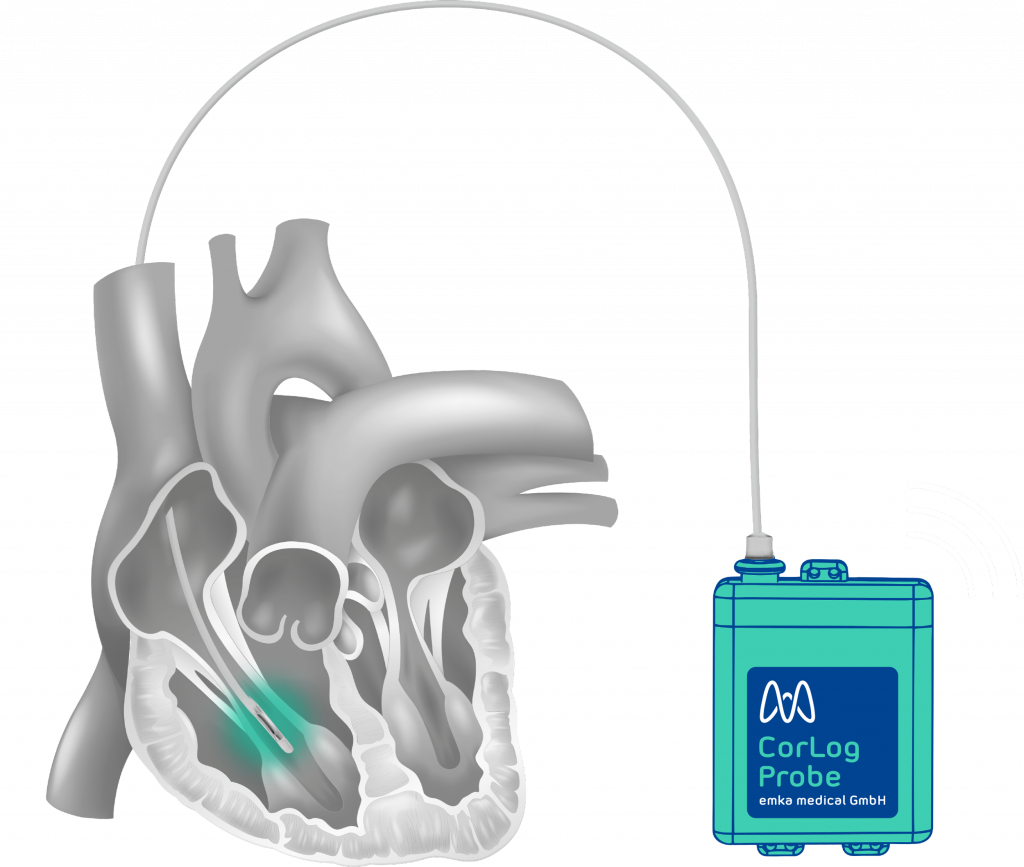

The CorLog System
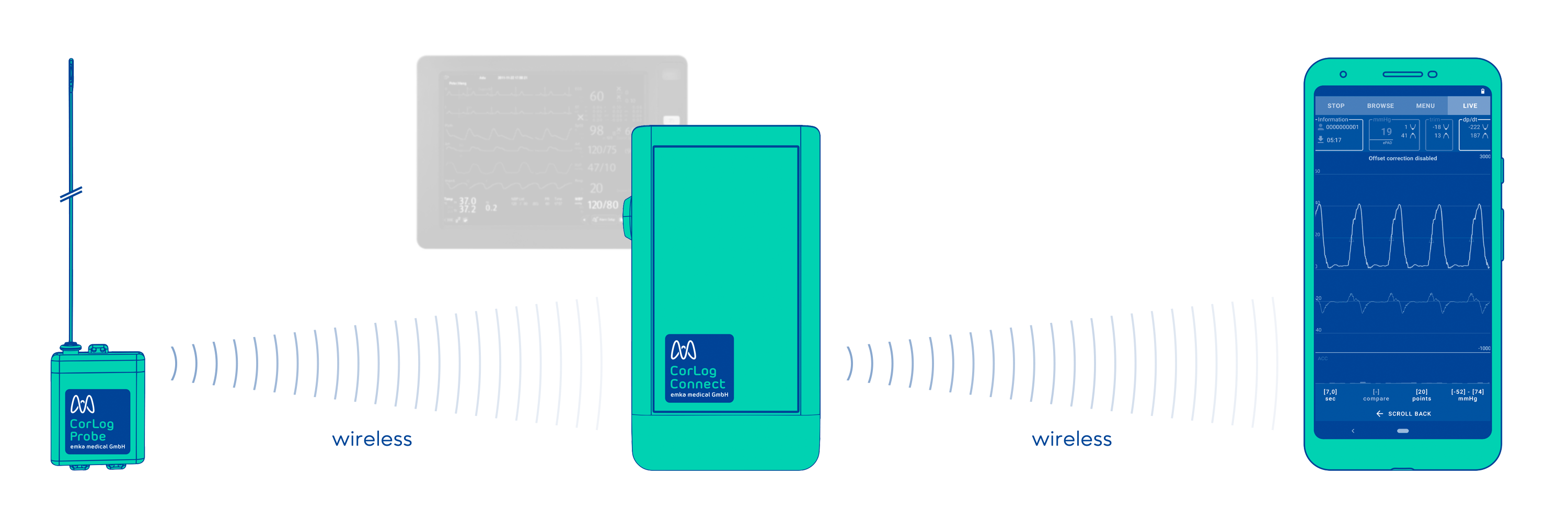
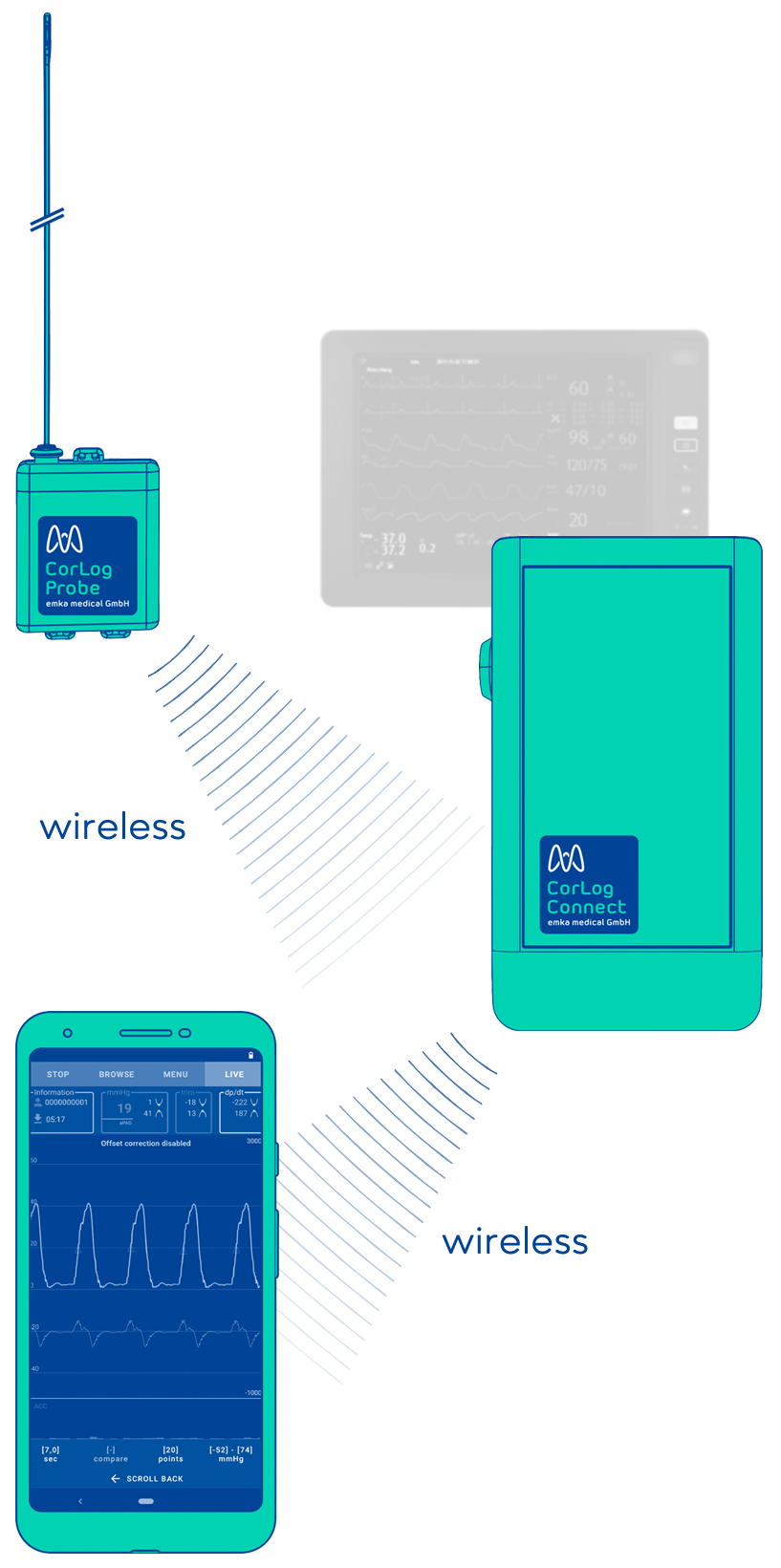
The use of CorLog
1. Prepare introduction set and split sheath
2. Insert CorLog Probe trough split sheath until right ventricle
3. Suture housing to the skin and add wound dressing
4. Plug in CorLog Connect and zero monitoring
5. CorLog is ready to go for up to 30 days
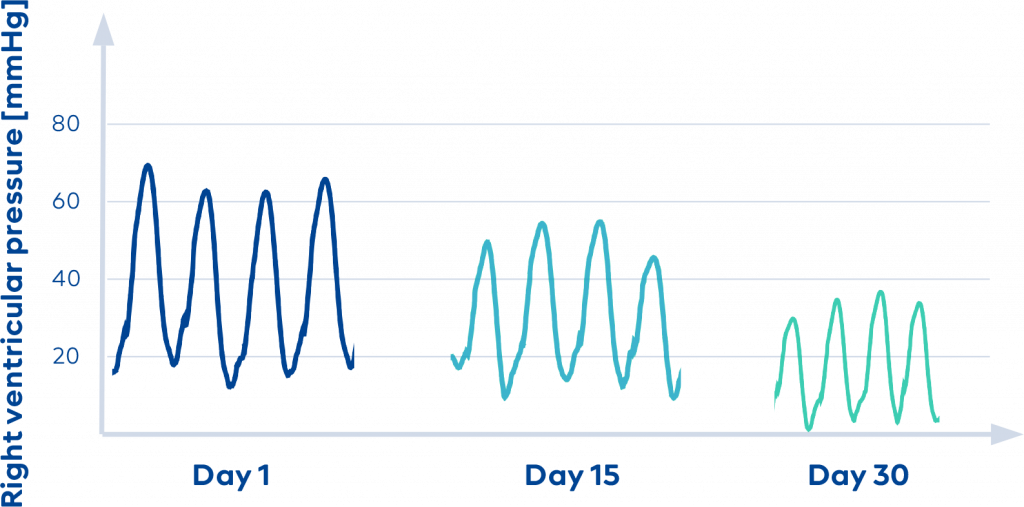
Why CorLog?
CorLog guides your therapy with the right data* continuously measured from the right heart. Live and unplugged from your heart failure patient.
Information when it matters.
*CorLog data can be used to continuously extract right ventricular ejection fraction and stroke volume.
The CorLog Devices

CorLog Probe
Electronics module, placed on the patient body, attached to measurement catheter.
CorLog Connect
Transmission module which can be connected to a standard patient monitor.
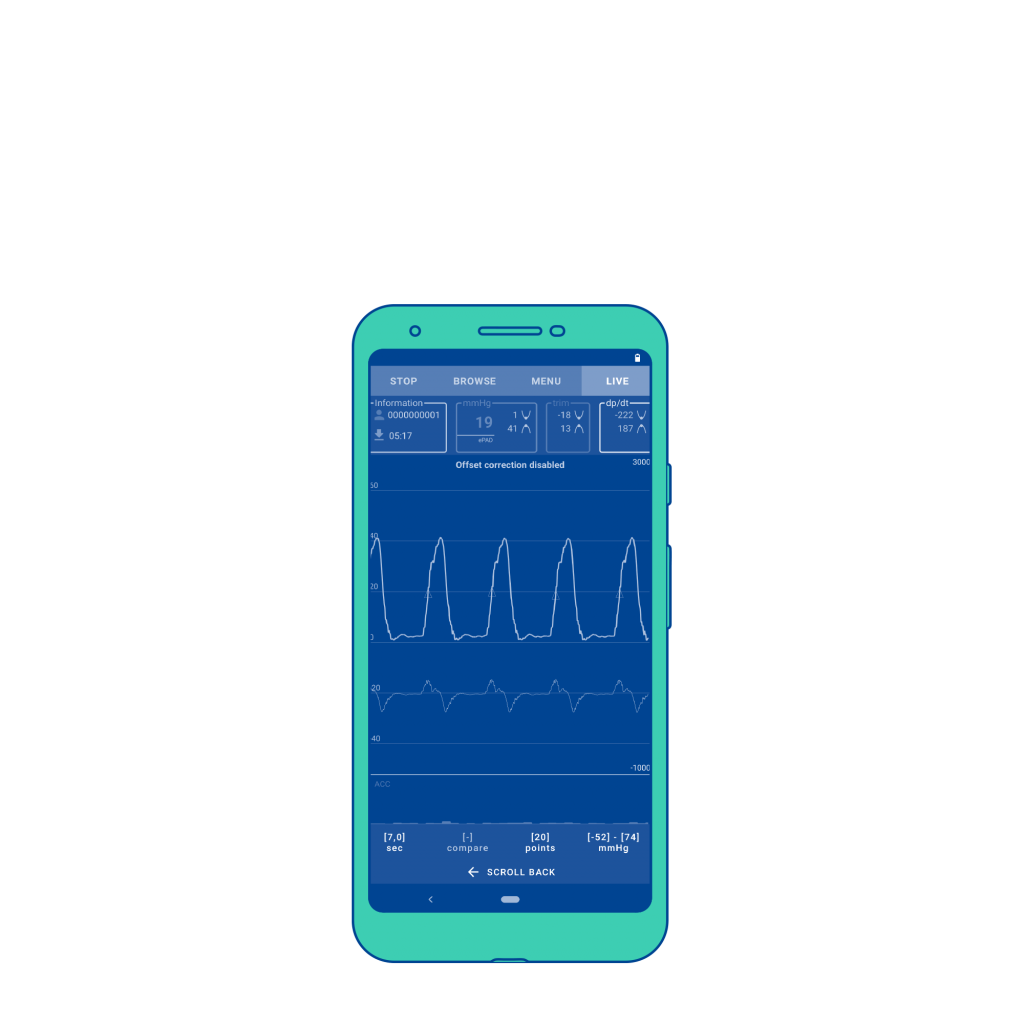
CorLog App
Displays and records invasive blood pressure signal from the right heart.

Get in touch
emka medical GmbH
Erthalstraße 12
63739 Aschaffenburg, Germany
P: +49 6021 4434522
F: +49 6021 4434523
E: info@emka-medical.de
Job applications:
bewerbung@emka-medical.de
Funding programs
emka medical has received the following grants and awards:
- Bavarian Minister of Economy and Technology BayMED
- Horizon 2020 SEAL of EXCELLENCE
© emka medical GmbH. All rights reserved. Legal Notice I Privacy Policy I Site by kw
